This aluminum battery uses a rechargeable mechanism that would replace existing storage devices like alkaline batteries and lithium-ion batteries, of which are bad for the environment and have been known to burst into flames, respectively. Hongjie Dai, a materials chemistry professor at Stanford,confident in the reliability of the new technology, states, "Our new battery won't catch fire, even if you drill through it."
The prototype is composed of both aluminum and graphite, allowing for a virtually indestructible hardware that continues to supply charge even after drilling into it, as has been tested in the lab, whereas other batteries might, as aforementioned, burst into flame.
These aluminum-ion batteries would succeed the lithium-ion batteries currently found in phones, computers, and tablets. The new battery would be less hazardous, and thus more environmentally friendly.
Aluminum is an ideal material because of its low flammability, low cost, and a high-charge storage capacity. The biggest deterrent until now has been finding the right parts that would allow for an aluminum-ion battery capable of successively producing a viable charge after repeated use. The new aluminum-ion prototype can withstand over 7,500 cycles without capacity loss, while previous attempts could only achieve 100 cycles and lithium-ion batteries around 1,000 cycles.
Aluminum is also quite bendable, and as such the materials could be manipulated to work for new technologies like bendable wearable smart devices.
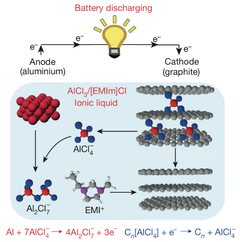
The aluminum-based battery is made up of a negatively-charged anode and a positively-charged cathode. "People have tried different kinds of materials for the cathode," Dai said. "We accidentally discovered that a simple solution is to use graphite, which is basically carbon. In our study, we identified a few types of graphite material that give us very good performance."
The aluminum anode and graphite cathode, in addition to an ionic-liquid-electrolyte, function within a flexible polymer-coated pouch. To top it off, "the electrolyte is basically a salt that's liquid at room temperature, so it's very safe," says Ming Gong, a graduate student conducting research within Dai’s lab.
Most importantly, these new aluminum-ion batteries could be used with electrical grids across the country, to store and release renewable energy rapidly over long periods of time. Plus, whereas these alkaline batteries are only able to create 1.5 volts of electricity, the new aluminum batteries can achieve 2.0 volts. However, lithium-ion batteries produce over twice as much voltage as does this new aluminum-based prototype, demonstrating that to effectively replace lithium-ion technology, the voltage of the current prototype will need to be improved.
"But improving the cathode material could eventually increase the voltage and energy density.” Dai said. “Otherwise, our battery has everything else you'd dream that a battery should have: inexpensive electrodes, good safety, high-speed charging, flexibility and long cycle life. I see this as a new battery in its early days. It's quite exciting."
-Bryan Díaz, Writer
 RSS Feed
RSS Feed

